Information about the chemistry behind TNT
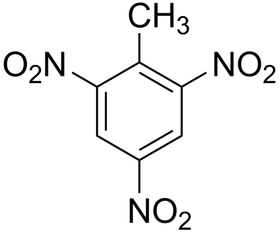
Formula: CH3C6H2(NO2)3
Structure: this compound is comprised of a Methyl and 3 nitro groups situated around a benzene ring.
Name: 2,4,6-Trinitrotoluene
Molar Mass: 277.01
Melting Point: 80.9 °C
Density: 1.654 g/cm3
Ignites: 259 °C
Energy: 4.184 megajoules per kilogram
Proporties:TNT is a yellowy clay like subastance. While extreemly combustable, TNT is able to withstand an extreme amount of shock and friction. One can litterally drive a hammer through a piece of dynamite and it will not detonate.Uses: TNT is one of the most well known explosives, it is commonly used in industrial demolition, and is used for military purposes
Synthesis: Toulene is nitrated to form mono-nitroluene. Then the mono-nitroluene is nitrated again to form Dinitrotoluene. then it is nitrated a third time with nitric acid and oleum. This process is how industries synthesise TNT
Structure: this compound is comprised of a Methyl and 3 nitro groups situated around a benzene ring.
Name: 2,4,6-Trinitrotoluene
Molar Mass: 277.01
Melting Point: 80.9 °C
Density: 1.654 g/cm3
Ignites: 259 °C
Energy: 4.184 megajoules per kilogram
Proporties:TNT is a yellowy clay like subastance. While extreemly combustable, TNT is able to withstand an extreme amount of shock and friction. One can litterally drive a hammer through a piece of dynamite and it will not detonate.Uses: TNT is one of the most well known explosives, it is commonly used in industrial demolition, and is used for military purposes
Synthesis: Toulene is nitrated to form mono-nitroluene. Then the mono-nitroluene is nitrated again to form Dinitrotoluene. then it is nitrated a third time with nitric acid and oleum. This process is how industries synthesise TNT
